Test Updates - HbA1c, FIB-4 Index, FT3
HbA1c: Updated Diagnostic Thresholds
The Diabetes National Clinical Network has issued updated HbA1c diagnostic thresholds to align New Zealand reporting with global standards and to support improved patient outcomes.
- Diabetes: HbA1c ≥ 48 mmol/mol (lowered from the current ≥ 50 mmol/mol).
- Prediabetes: HbA1c 42 – 47 mmol/mol (increased from the current 40 mmol/mol).
- Normal: HbA1c < 42 mmol/mol.
- A confirmatory test will not be required where the initial HbA1c is > 53 mmol/mol.
HbA1c result flagging will occur if the result is 42 mmol/mol or above (currently this is 40 mmol/mol). In addition, explanatory comments will be added depending on the HbA1c result (as provided by the Diabetes National Clinical Network).
The above changes will be implemented by Pathlab on Wednesday 22nd April 2026, ahead of the Ministry of Health deadline of 1st July 2026.
Please see our website for a complete list of HbA1c levels and applicable comments: https://tests.pathlab.co.nz/tests/hba1c/
FIB-4 Index (Liver Fibrosis Score)
Pathlab is now able to offer FIB-4 Index as a requestable test.
The FIB-4 Index is used to assess the risk of liver fibrosis in patients with Metabolic Associated Fatty Liver Disease (but is not appropriate for other causes of liver disease, see https://www.rcpa.edu.au/Manuals/RCPA-Manual/Pathology-Tests/F/FIB-4).
FIB-4 Index is a calculation which uses the patient’s age, and AST, ALT, and platelet results. Please see our website for FIB-4 limits and comments, which have been adopted from the Auckland Region Gastroenterology group: https://tests.pathlab.co.nz/tests/fib-4-index/
If using Pathlab Éclair orders, please go to the Additional tab and search "FIB-4."
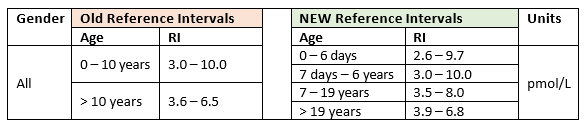
FT3 Reference Intervals
In consultation with Endocrinologists and Paediatricians, Pathlab has recently updated reference intervals for FT3. This aligns Pathlab with Waikato Hospital Laboratory and LabPlus and includes more appropriate intervals for young paediatric patients. A summary is listed below.
These new reference intervals have been in effect from Thursday 5th March 2026. Results that fall outside the reference interval continue to be flagged. Please see our website for a full list of reference intervals, including the updated FT3 intervals: https://www.pathlab.co.nz/providers